Centre for International Accreditation (CIA)
Call Us when you Need Help!
About CIA
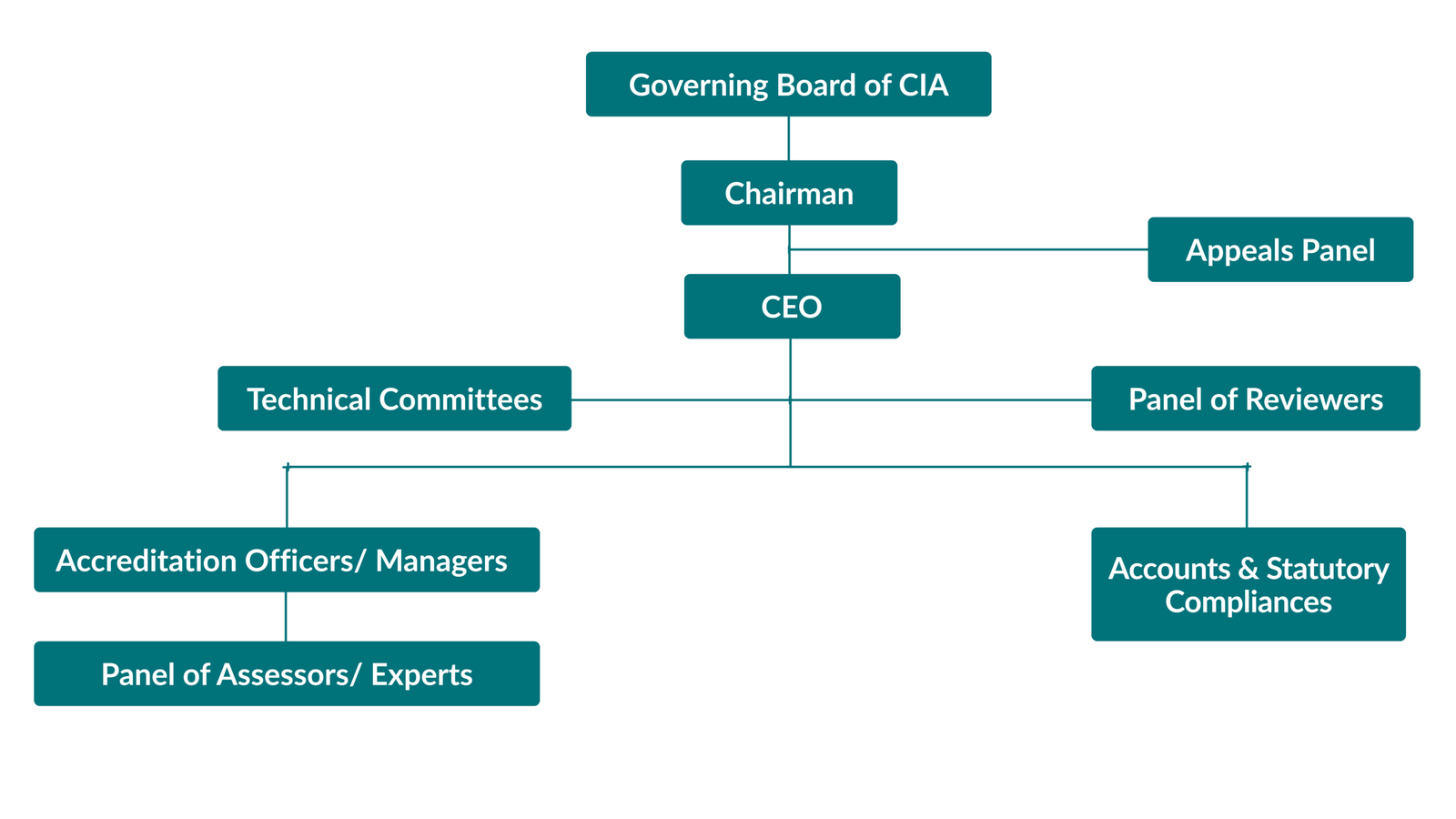
IntroductionQAI’s CIA has been established with the objective of providing Government, Industry Associations and Industry in general with a scheme of accreditation of Conformity Assessment Bodies (CABs) as per the international standards such as medical testing laboratories as per ISO 15189; testing and calibration laboratories as per ISO/IEC 17025; biobanking as per ISO 20387; proficiency testing provider as per ISO/IEC 17043; reference material producers as per ISO 17034; and inspection bodies as per ISO/IEC 17020. We offer accreditation services in a non-discriminatory manner. These services are accessible to all qualified CABs in India and other countries regardless of the size, its membership of any association or group. QAI CIA has established its accreditation system in accordance with ISO/ IEC 17011 ‘Conformity Assessment – General requirements for accreditation bodies accrediting conformity assessment bodies and Asia Pacific Accreditation Cooperation (APAC). QAI CIA is a full member/ signatory to the Mutual Recognition Arrangements (MRA) of the APAC and International Laboratory Accreditation Programme (ILAC) (which will be transitioned to the Global Accreditation Cooperation Incorporated effective 1 January 2026). The organization structure of CIA has been designed to meet the requirements of an effective and efficient accreditation system. The Centre is governed by a Board. The Board frames the policies and guidelines, and provide direction. CEO, QAI is the Member Secretary of the Board. CIA operates its accreditation process through a structured framework of competent staff, a pool of empanelled Lead Assessors and Assessors, and various committees covering specified expertise. |
QAI CIA has achieved global recognition through APAC and ILAC MRA in Testing and Calibration (ISO/IEC 17025) and Medical (ISO 15189).
APAC Mutual Recognition Arrangement (MRA) Certificate

ILAC Mutual Recognition Arrangement (MRA) Certificate

Quality PolicyCentre for International Accreditation’ of Quality and Accreditation Institute (QAI) will provide accreditation service to the satisfaction of its customers in accordance with the national and international norms. It is committed to provide equal opportunity to all the applicants with regard to impartiality, transparency, integrity, and confidentiality. The Centre will strive for the international recognition of its accreditation programmes through regional and international forums like APAC, ILAC etc. and through bilateral and multilateral mutual recognition arrangements. We aim to operate globally. We strive to comply with the requirements of ISO/IEC 17011:2017 “Conformity assessment —requirements for accreditation bodies accrediting conformity assessment bodies” and other relevant national/international standards and applicable APAC/ILAC documents. |
Impartiality PolicyImpartiality is the backbone of QAI CIA activities and is of paramount significance in maintaining the confidence amongst the stakeholders about its accreditation system. QAI CIA is committed to safeguard the impartiality while dealing with accreditation at all levels. QAI CIA adheres to the requirement of ISO/IEC 17011: 2017 ‘Conformity assessment – Requirements for accreditation bodies accrediting conformity assessment bodies’ that accreditation shall be undertaken impartially. |
Vision:
Nurturing the largest global pool of organisations and people through quality and accreditation framework.
Mission:
To conceive and deliver education, training, accreditation and related programmes in partnership with stakeholders using an approach of co-design and co-creation.
Our Core Values:
Listener: Seek continuous feedback from stakeholders to address their concerns
Competitive: Look for viable options to benefit users of our services
Transparency: Clearly defined policies made available in public domain
Innovation: Continuously evolve using co-design and co-creation
ScopeCentre for International Accreditation (CIA) provides accreditation to following types of Conformity Assessment Bodies (CABs):Impartiality is the backbone of QAI CIA activities and is of paramount significance in maintaining the confidence amongst the stakeholders about its accreditation system. QAI CIA is committed to safeguard the impartiality while dealing with accreditation at all levels. QAI CIA adheres to the requirement of ISO/IEC 17011: 2017 ‘Conformity assessment – Requirements for accreditation bodies accrediting conformity assessment bodies’ that accreditation shall be undertaken impartially.
|
It also offers recognition scheme for medical labs as per clinical establishment act requirements.
Board – Centre for International Accreditation (CIA)
Member

Shobhna Sharma
Member

Dr. N. V. Ramarao
Member

Ajay Soni
Organisation Structure for Laboratory Accreditation (CIA)
CIA General Documents
Medical Laboratory Accreditation
| S.NO. | CERTIFICATE NUMBER | NAME OF MEDICAL LABORATORY | CITY/COUNTRY | VALID FROM | VALID UNTIL |
| 1. | QAI/CIA/ML/2019/0002 | NARAYANA NETHRALAYA INSTITUTE OF MOLECULAR DIAGNOSTICS AND LABORATORY SERVICES - I | BENGALURU/INDIA | FEB 18, 2025 | FEB 17, 2027 |
| 2. | QAI/CIA/ML/2019/0003 | NARAYANA NETHRALAYA INSTITUTE OF MOLECULAR DIAGNOSTICS AND LABORATORY SERVICES - II | BENGALURU/INDIA | FEB 18, 2025 | FEB 17, 2027 |
| 3. | QAI/CIA/ML/2022/0013 | EASTERN REGIONAL HOSPITAL LABORATORY | KOFORIDUA/GHANA | AUG 26, 2025 | AUG 25 ,2027 |
| 4. | QAI/CIA/ML/2022/0014 | GREATER ACCRA REGIONAL HOSPITAL LABORATORY | ACCRA/GHANA | SEP 27, 2024 | SEP 26, 2026 |
| 5. | QAI/CIA/ML/2023/0015 | LAB SCIENCE DIAGNOSTICS | DHAKA/BANGLADESH | NOV 03, 2025 | NOV 02, 2027 |
| 6. | QAI/CIA/ML/2023/0017 | PRATHAM PATHOLOGY LABORATORY PRIVATE LIMITED | KATHMANDU/ NEPAL | APR 06, 2026 | APR 05, 2028 |
| 6. | QAI/CIA/ML/2024/0020 | CENTRE OF MEDICAL LABORATORY (MONGOLIA-JAPAN HOSPITAL OF THE MONGOLIAN NATIONAL UNIVERSITY OF MEDICAL SCIENCES) | ULAANBAATAR/MONGOLIA | JUN 30, 2024 | JUN 29, 2026 |
| 7. | QAI/CIA/ML/2024/0021 | AMARA RAJA OCCUPATIONAL HEALTH CENTER MEDICAL LABORATORY (A UNIT OF AMARA RAJA ENERGY & MOBILITY LTD.) | TIRUPATI/INDIA | JUL 08, 2024 | JUL 07, 2026 |
| 8. | QAI/CIA/ML/2024/0022 | AMARA RAJA OCCUPATIONAL HEALTH CENTER MEDICAL LABORATORY (A UNIT OF AMARA RAJA ENERGY & MOBILITY LTD.) | CHITTOOR/INDIA | JUL 08, 2024 | JUL 07, 2026 |
| 9. | QAI/CIA/ML/2024/0023 | URO LAB (A DIVISION OF STERLING ACCURIS WELLNESS PVT. LTD.) | AHMEDABAD/ INDIA | OCT 04, 2024 | OCT 03, 2026 |
| 10. | QAI/CIA/ML/2024/0024 | CENTRAL DIAGNOSTIC LAB | KATHMANDU/NEPAL | NOV 12, 2024 | NOV 11, 2026 |
| 11. | QAI/CIA/ML/2024/0025 | BIODIAGNOSTIC PATH LABS NEPAL PVT. LTD. | KATHMANDU/NEPAL | NOV 12, 2024 | NOV 11, 2026 |
| 12. | QAI/CIA/ML/2024/0026 | VASAVI DIAGNOSTIC LABORATORY | CHITRADURGA/INDIA | NOV 23, 2024 | NOV 22, 2026 |
| 13. | QAI/CIA/ML/2024/0027 | SIGMA DIAGNOSTICS LABORATORY PVT. LTD. | BHAKTAPUR/NEPAL | DEC 10, 2024 | DEC 09, 2026 |
| 14. | QAI/CIA/ML/2025/0028 | NIRAMAYA DIAGNOSTICS PVT. LTD. | KATHMANDU/NEPAL | MAR 25, 2025 | MAR 24,2027 |
| 15. | QAI/CIA/ML/2025/0029 | BAHRAIN MEDICAL LABORTORY | BAHRAIN | JUN 19, 2025 | JUN 18 ,2027 |
| 16. | QAI/CIA/ML/2025/0030 | MEDBIOTICA DIAGNOSTIC CENTRE | VISAKAPATNMAM/NDIA | SEP 23 , 2025 | SEP 22, 2027 |
| 17. | QAI/CIA/ML/2025/0031 | REXPRESS HEALTH SOLUTIONS PRIVATE LIMITED (MOBILE MEDICAL LABORATORY) | DELHI/ INDIA | OCT 04, 2025 | OCT 03, 2027 |
| 18. | QAI/CIA/ML/2026/0032 | REXPRESS HEALTH SOLUTIONS PRIVATE LIMITED (MOBILE MEDICAL LABORATORY) | DELHI/ INDIA | MAR 09, 2026 | MAR 08, 2028 |
| SL.NO. | CERTIFICATE NO. | NAME OF THE CAB | CITY/COUNTRY | VALID FROM | VALID UNTIL |
| 1. | QAI/CIA/ML/2023/0016 | SYNERGY CARE CLINIC OF PROSYNERGY MEDICAL SYSTEMS PVT LTD. | HULHUMALE/ REPUBLIC OF MALDIVES | SEP 22, 2023 | SEP 21, 2025 |
| 2. | QAI/CLA/ML/2018/0001 | DOCTOR.E LABORATORY | NEW DELHI/ INDIA | MAR 26, 2018 | MAR 25, 2020 |
| 3. | QAI/CLA/ML/2019/0004 | ELOI DIAGNOSTICS | ERNAKULAM/INDIA | JUN 04, 2019 | JUN 03, 2021 |
| 4. | QAI/CLA/ML/2019/0008 | BENGALURU GENOMICS CENTRE | BENGALURU/INDIA | OCT 15, 2019 | OCT 14, 2021 |
| 5. | QAI/CLA/ML/2019/0009 | RAJIV GANDHI CENTRE FOR BIOTECHNOLOGY, MEDICAL LABORATORY SERVICES, MEDICAL COLLEGE HOSPITAL | THIRUVANANTHAPURAM/INDIA | DEC 04, 2019 | 03 DEC, 2021 |
| 6. | QAI/CLA/ML/2019/0010 | RAJIV GANDHI CENTRE FOR BIOTECHNOLOGY, MEDICAL LABORATORY SERVICES, GENERAL HOSPITAL | THIRUVANANTHAPURAM/INDIA | DEC 02, 2019 | DEC 01, 2021 |
| 7. | QAI/CLA/ML/2019/0011 | RAJIV GANDHI CENTRE FOR BIOTECHNOLOGY, MEDICAL LABORATORY SERVICES, DISTRICT HOSPITAL | THIRUVANANTHAPURAM/INDIA | DEC 04, 2019 | DEC 03, 2021 |
| 8. | QAI/CLA/ML/2020/0012 | KANVA DIAGNOSTIC SERVICES PVT. LTD. | BENGALURU/INDIA | JAN 06, 2022 | JAN 05, 2024 |
| 10. | QAI/CLA/ML/2023/0017 | PRATHAM PATHOLOGY LABORATORY PRIVATE LIMITED | KATHMANDU/NEPAL | OCT 31, 2023 | OCT 30, 2025 |
Testing Laboratory Accreditation
| S. No. | Accreditation Certificate No. | Name of Accredited Testing Laboratories | City/Country | Valid From | Valid until |
| 1. | QAI/CIA/TL/2019/0002 | VALIDAIR ENGINEERS | AHMEDABAD/INDIA | FEB 24, 2026 | FEB 23, 2028 |
| 2. | QAI/CIA/TL/2020/0003 | KVAFSU-CVA RABIES DIAGNOSTIC LABORATORY | BENGALURU/INDIA | FEB 08, 2026 | FEB 07, 2028 |
| 3. | QAI/CIA/TL/2023/0005 | JBS TESTING SOLUTIONS (UNIT 2) | JALANDHAR/INDIA | FEB 13, 2025 | FEB 12, 2027 |
| 4. | QAI/CIA/TL/2023/0006 | PLANET ELECTRO LABS PRIVATE LIMITED | NEW DELHI/INDIA | FEB 16, 2025 | FEB 15, 2027 |
| 5. | QAI/CIA/TL/2023/0008 | PERFICIO TEST AND RESEARCH CENTRE | VADODARA/ INDIA | APR 12, 2025 | APR 11, 2027 |
| 6. | QAI/CIA/TL/2023/0009 | IEC TEST LABS LLP | DELHI/INDIA | APR 29, 2025 | APR 28, 2027 |
| 7. | QAI/CIA/TL/2023/0010 | SLEEN INDIA BIZVENTURE PVT. LTD. | AGRA/INDIA | MAY 25, 2025 | MAY 24, 2027 |
| 8. | QAI/CIA/TL/2023/0011 | ARDM LABS PRIVATE LIMITED | DELHI/INDIA | JUL 10, 2025 | JUL 09, 2027 |
| 9. | QAI/CIA/TL/2023/0012 | BUREAU VERITAS CONSUMER PRODUCT SERVICES (INDIA) PVT. LTD. | BENGALURU/INDIA | JUN 20, 2025 | JUN 19, 2027 |
| 10. | QAI/CIA/TL/2023/0013 | MITRA S. K. PRIVATE LIMITED | BHUBANESWAR/INDIA | JUL 12, 2025 | JUL 11, 2027 |
| 11. | QAI/CIA/TL/2023/0014 | SHIVA TEST HOUSE PRIVATE LIMITED | GHAZIABAD/INDIA | JUL 12, 2025 | JUL 11, 2027 |
| 12. | QAI/CIA/TL/2023/0015 | MITRA S. K. PRIVATE LIMITED | KOLKATA/INDIA | JUL 13, 2025 | JUL 12, 2027 |
| 13. | QAI/CIA/TL/2023/0017 | AADITECH TEST AND CALIBRATION LAB LLP | NEW DELHI/INDIA | JUL 24, 2025 | JUL 23, 2027 |
| 14. | QAI/CIA/TL/2023/0018 | INTERTEK INDIA PRIVATE LIMITED | MUMBAI/INDIA | JUL 24, 2025 | JUL 23, 2027 |
| 15. | QAI/CIA/TL/2023/0021 | INTERTEK INDIA PRIVATE LIMITED | BENGALURU/INDIA | AUG 09, 2025 | AUG 08, 2027 |
| 16. | QAI/CIA/TL/2023/0022 | PRECISION LABORATORIES LLP | AHMEDABAD/INDIA | AUG 31, 2025 | AUG 30, 2027 |
| 17. | QAI/CIA/TL/2023/0023 | HT PRODUCT SERVICES PRIVATE LIMITED (FORMERLY HOUSE OF TESTING) | NOIDA/INDIA | FEB 03, 2026 | FAB 02, 2028 |
| 18. | QAI/CIA/TL/2023/0024 | WELL TECH TEST AND RESEARCH CENTER | MOHALI/INDIA | NOV 21, 2025 | NOV 20, 2027 |
| 19. | QAI/CIA/TL/2023/0025 | INTERTEK INDIA PRIVATE LIMITED | CHENNAI/INDIA | SEP 15, 2025 | SEP 14, 2027 |
| 20. | QAI/CIA/TL/2023/0026 | VENTURE GULF UNITECH METALLURGICAL SERVICES W.L.L | DOHA/QATAR | SEP 27, 2025 | SEP 26, 2027 |
| 21. | QAI/CIA/TL/2023/0027 | MITRA S. K. PRIVATE LIMITED | KATNI/INDIA | OCT 07, 2025 | OCT 06, 2027 |
| 22. | QAI/CIA/TL/2023/0028 | MIET ANIMAL HOUSE FACILITY & MIET CELL CULTURE LAB (MEERUT INSTITUTE OF ENGINEERING AND TECHNOLOGY, CITY EDUCATIONAL AND SOCIAL WELFARE SOCIETY) | MEERUT/INDIA | OCT 10, 2025 | OCT 09, 2027 |
| 23. | QAI/CIA/TL/2023/0029 | UNIVERSAL TESTING AND RESEARCH CENTRE (A DIVISION OF SUSTAINABLE STEWARDSHIP PRIVATE LIMITED) | NEW DELHI/INDIA | OCT 26, 2025 | OCT 25, 2027 |
| 24. | QAI/CIA/TL/2023/0030 | FINE FINISH ANALYTICAL LABORATORY (A DIVISION OF FINE FINISH ORGANICS PRIVATE LIMITED) | RAIGAD/INDIA | OCT 28, 2025 | OCT 27, 2027 |
| 25. | QAI/CIA/TL/2023/0031 | ATMY ANALYTICAL LABS PVT. LTD. | GREATER NOIDA/INDIA | NOV 11, 2025 | NOV 10, 2027 |
| 26. | QAI/CIA/TL/2023/0032 | URS PRODUCTS AND TESTING PRIVATE LIMITED | NOIDA/INDIA | NOV 17, 2025 | NOV 16, 2027 |
| 27. | QAI/CIA/TL/2023/0033 | TVS LABS | DELHI/INDIA | NOV 25, 2025 | NOV 24, 2027 |
| 28. | QAI/CIA/TL/2023/0034 | BUREAU VERITAS CONSUMER SERVICES (BD) LTD. | DHAKA/BANGLADESH | DEC 01, 2025 | NOV 30, 2027 |
| 29. | QAI/CIA/TL/2024/0036 | MASTER LABS FOR TECHNICAL TESTING, INSPECTION AND CALIBRATION SERVICES | HIDD/ KINGDOM OF BAHRAIN | JAN 6, 2024 | JAN 5, 2026 |
| 30. | QAI/CIA/TL/2024/0037 | AJEO TESTING LABS PRIVATE LIMITED | GHAZIABAD/INDIA | FEB 02, 2026 | FEB 01, 2028 |
| 31. | QAI/CIA/TL/2024/0038 | COTECNA INSPECTION INDIA PRIVATE LIMITED | GURUGRAM/ INDIA | FEB 02, 2026 | FEB 01, 2028 |
| 32. | QAI/CIA/TL/2024/0039 | AIKANTIKA TESTING CENTRE | GHAZIABAD/INDIA | FEB 10, 2024 | FEB 09, 2026 |
| 33. | QAI/CIA/TL/2024/0040 | COTECNA INSPECTION INDIA PRIVATE LIMITED | VISAKHAPATNAM/ INDIA | FEB 15, 2024 | FEB 14, 2026 |
| 34. | QAI/CIA/TL/2024/0041 | COTECNA INSPECTION INDIA PRIVATE LIMITED | PURBA MEDINIPUR/ INDIA | FEB 15, 2024 | FEB 14, 2026 |
| 35. | QAI/CIA/TL/2024/0042 | COTECNA INSPECTION INDIA PRIVATE LIMITED | KUTCH/ INDIA | FEB 17, 2024 | FEB 16, 2026 |
| 36. | QAI/CIA/TL/2024/0043 | COTECNA INSPECTION INDIA PRIVATE LIMITED | CHENNAI/ INDIA | FEB 17, 2024 | FEB 16, 2026 |
| 37. | QAI/CIA/TL/2024/0045 | INTERTEK INDIA PRIVATE LIMITED | BENGALURU/INDIA | FEB 22, 2024 | FEB 21, 2026 |
| 38. | QAI/CIA/TL/2024/0046 | EPYLLION TESTING LAB LTD. | GAZIPUR/ BANGLADESH | FEB 26, 2024 | FEB 25, 2026 |
| 39. | QAI/CIA/TL/2024/0047 | CLASSIC TESTING & RESEARCH CENTRE | NOIDA/ INDIA | MAR 08, 2024 | MAR 07, 2026 |
| 40. | QAI/CIA/TL/2024/0048 | JBS TESTING SOLUTIONS PRIVATE LIMITED | JALANDHAR/INDIA | APR 09,2024 | APR 08,2026 |
| 41. | QAI/CIA/TL/2024/0049 | BUREAU VERITAS CONSUMER PRODUCTS (INDIA) PVT. LTD. | NOIDA/INDIA | APR 10, 2024 | APR 09, 2026 |
| 42. | QAI/CIA/TL/2024/0050 | CLASSIC INSTRUMENTATION PVT. LTD. | NOIDA/ INDIA | MAY 10, 2024 | MAY 09, 2026 |
| 43. | QAI/CIA/TL/2024/0051 | EUROASIA SCIENTIFIC INDIA PVT. LTD. | GURUGRAM/ INDIA | MAY 17, 2024 | MAY 16, 2026 |
| 44. | QAI/CIA/TL/2024/0052 | VARDAN ENVIROLAB LLP | GURUGRAM/ INDIA | MAY 29, 2024 | MAY 28, 2026 |
| 45. | QAI/CIA/TL/2024/0053 | GOLDEN DOWA ECO-SYSTEM MYANMAR CO LTD. | YANGON/ MYANMAR | MAY 30, 2024 | MAY 29 , 2026 |
| 46. | QAI/CIA/TL/2024/0054 | U KAR KA FOOD TESTING LABORATORY (A UNIT OF U KAR KA COMPANY LIMITED) | MANDALAY/ MYANMAR | MAY 31, 2024 | MAY 30, 2026 |
| 47. | QAI/CIA/TL/2024/0055 | QA TESTING LABORTORY PVT. LTD. | NOIDA/ INDIA | MAY 31, 2024 | MAY 30, 2026 |
| 48. | QAI/CIA/TL/2024/0056 | BUREAU VERITAS CONSUMER PRODUCTS SERVICES (CTG) LTD. | CHITTAGONG/ BANGLADESH | MAY 31, 2024 | MAY 30, 2026 |
| 49. | QAI/CIA/TL/2024/0057 | BUNITECH METALLURGICAL SERVICES PVT. LTD. | AHMEDABAD/ INDIA | MAY 31, 2024 | MAY 30, 2026 |
| 50. | QAI/CIA/TL/2024/0058 | QA TESTING LABORTORY PVT. LTD. | KORBA/ INDIA | JUN 08, 2024 | JUN 07, 2026 |
| 51. | QAI/CIA/TL/2024/0059 | ELUTECH LABORATORY AND CALIBRATION SERVICES | JAIPUR/ INDIA | JUN 12, 2024 | JUN 11, 2026 |
| 52. | QAI/CIA/TL/2024/0060 | BLADE TEST CENTRE GUJARAT (A UNIT OF SUZLON ENERGY LIMITED) | VADODARA/ INDIA | JUN 19, 2024 | JUN 18, 2026 |
| 53. | QAI/CIA/TL/2024/0061 | COTECNA INSPECTION INDIA PRIVATE LIMITED | PARADEEP/ INDIA | JUN 19, 2024 | JUN 18, 2026 |
| 54. | QAI/CIA/TL/2024/0062 | EUROFINS CONSUMER PRODUCT TESTING INDIA PRIVATE LIMITED (FORMELY MODERN TESTING SERVICES (INDIA) PRIVATE LIMITED) | NOIDA/ INDIA | JUN 28, 2024 | JUN 27, 2026 |
| 55. | QAI/CIA/TL/2024/0063 | ALAIPURIA TEST HOUSE PRIVATE LIMITED | GHAZIABAD/INDIA | JUL 02, 2024 | JUL 01, 2026 |
| 56. | QAI/CIA/TL/2024/0064 | QUALITY SERVICES & SOLUTIONS PRIVATE LIMITED | DHANBAD/ INDIA | JUL 08, 2024 | JUL 07, 2026 |
| 57. | QAI/CIA/TL/2024/0065 | GEO CHEM CONSUMER PRODUCTS SERVICES (CPS) LTD. | DHAKA/ BANGLADESH | JUL 11, 2024 | JUL 10, 2026 |
| 58. | QAI/CIA/TL/2024/0066 | EMC TESTING AND COMPLIANCE LLP | GURUGRAM/ INDIA | JUL 17, 2024 | JUL 16, 2026 |
| 59. | QAI/CIA/TL/2024/0067 | NATIONAL TESTING BUREAU | GHAZIABAD/INDIA | JUL 26, 2024 | JUL 25, 2026 |
| 60. | QAI/CIA/TL/2024/0068 | BHARAT VANIJYA EASTERN PRIVATE LIMITED TESTING LABORATORY | PALAMU/ INDIA | JULY 29, 2024 | JULY 28, 2026 |
| 61. | QAI/CIA/TL/2024/0069 | BHARAT VANIJYA EASTERN PRIVATE LIMITED TESTING LABORATORY | RAICHUR/ INDIA | AUG 02, 2024 | AUG 01, 2026 |
| 62. | QAI/CIA/TL/2024/0070 | ACCURATE TEST SOLUTIONS LLP | NOIDA/ INDIA | AUG 06, 2024 | AUG 05, 2026 |
| 63. | QAI/CIA/TL/2024/0071 | NATURAL RUBBER TESTING LABORATORY | YANGON/ MYANMAR | AUG 06, 2024 | AUG 05, 2026 |
| 64. | QAI/CIA/TL/2024/0072 | AANKAN CALIBRATION AND TESTING CENTRE LLP | GANDHINAGAR/ INDIA | AUG 14, 2024 | AUG 13, 2026 |
| 65. | QAI/CIA/TL/2024/0073 | QUALITY SERVICES AND SOLUTIONS PRIVATE LIMITED | RAIGARH/ INDIA | AUG 16, 2024 | AUG 15, 2026 |
| 66. | QAI/CIA/TL/2024/0074 | EUROFINS CONSUMER PRODUCT TESTING INDIA PRIVATE LIMITED (FORMELY MODERN TESTING SERVICES (INDIA) PRIVATE LIMITED) | TIRUPUR/ INDIA | AUG 29, 2024 | AUG 28, 2026 |
| 67. | QAI/CIA/TL/2024/0075 | INDIAN TESTING LABORATORY PRIVATE LIMITED (FORMERLY INDIAN TESTING LABORATORY) | GREATER NOIDA/ INDIA | SEP 11, 2024 | SEP 10, 2026 |
| 68. | QAI/CIA/TL/2024/0076 | ENERGON SOLUTIONS PRIVATE LIMITED | NASHIK/ INDIA | SEP 12, 2024 | SEP 11, 2026 |
| 69. | QAI/CIA/TL/2024/0077 | MITRA S. K. PRIVATE LIMITED | GANDHIDHAM/ INDIA | SEP 14, 2024 | SEP 13, 2026 |
| 70. | QAI/CIA/TL/2024/0078 | AADVIK NATIONAL RESEARCH & TEST CENTER | KARNAL/ INDIA | SEP 28, 2024 | SEP 27, 2026 |
| 71. | QAI/CIA/TL/2024/0079 | EUROFINS CONSUMER PRODUCT TESTING INDIA PRIVATE LIMITED (FORMELY MODERN TESTING SERVICES (INDIA) PRIVATE LIMITED) | BENGALURU/INDIA | OCT 10, 2024 | OCT 09, 2026 |
| 72. | QAI/CIA/TL/2024/0080 | SHAURYA STEEL PTY. LTD. QA LAB | GAUTENG/SOUTH AFRICA | OCT 17, 2024 | OCT 16, 2026 |
| 73. | QAI/CIA/TL/2024/0081 | SHRI KRISHNA TEST HOUSE | DELHI/INDIA | OCT 22, 2024 | OCT 21, 2026 |
| 74. | QAI/CIA/TL/2024/0082 | QUALITY CONTROL LABORATORY (INDIAN HERBS SPECIALITIES PRIVATE LIMITED) | SAHARANPUR/INDIA | OCT 29, 2024 | OCT 28, 2026 |
| 75. | QAI/CIA/TL/2024/0083 | SAMRIDHI TEST HOUSE PRIVATE LIMITED | DELHI/INDIA | OCT 29, 2024 | OCT 28, 2026 |
| 76. | QAI/CIA/TL/2024/0084 | ATHARVA LABORATORIES PVT. LTD. | NOIDA/ INDIA | NOV 05, 2024 | NOV 04, 2026 |
| 77. | QAI/CIA/TL/2024/0085 | LABORATORY DIVISION, COTECNA INSPECTION INDIA SERVICES PRIVATE LIMITED | NEW DELHI/INDIA | NOV 05, 2024 | NOV 04, 2026 |
| 78. | QAI/CIA/TL/2024/0086 | SWASTIK ELECTRONICS TESTING CENTRE (OPC) PRIVATE LIMITED | GHAZIABAD/INDIA | NOV 08, 2024 | NOV 07, 2026 |
| 79. | QAI/CIA/TL/2024/0087 | ALPHA TEST HOUSE SERVICES LLP | MOHALI/INDIA | NOV 22, 2024 | NOV 21, 2026 |
| 80. | QAI/CIA/TL/2024/0088 | ASTUTE LABS PRIVATE LIMITED | PUNE/INDIA | NOV 26, 2024 | NOV 25, 2026 |
| 81. | QAI/CIA/TL/2024/0089 | MITRA S. K. PRIVATE LIMITED | VISAKHAPATNAM/INDIA | DEC 16, 2024 | DEC 15, 2026 |
| 82. | QAI/CIA/TL/2024/0090 | COTECNA INSPECTION INDIA PRIVATE LIMITED | TIRUPPUR/INDIA | DEC 20, 2024 | DEC 19, 2026 |
| 83. | QAI/CIA/TL/2024/0091 | ABNS SCIENTIFIC SERVICES | GUWAHATI/INDIA | DEC 20, 2024 | DEC 19, 2026 |
| 84. | QAI/CIA/TL/2024/0092 | MITRA S. K. PRIVATE LIMITED | BHUBANESWAR/ INDIA | DEC 24, 2024 | DEC 23, 2026 |
| 85. | QAI/CIA/TL/2024/0093 | TESTING LABORATORY, RAJ WOOLLEN INDUSTRIES | PANIPAT/INDIA | DEC 27, 2024 | DEC 26, 2026 |
| 86. | QAI/CIA/TL/2024/0094 | MITRA S. K. PRIVATE LIMITED | KOLKATA/INDIA | DEC 31, 2024 | DEC 30, 2026 |
| 87. | QAI/CIA/TL/2025/0095 | BASIL QUALITY TESTING LAB PRIVATE LIMITED | GURUGRAM/INDIA | JAN 02, 2025 | JAN 01, 2027 |
| 88. | QAI/CIA/TL/2025/0096 | DASHMESH TEST LAB PRIVATE LIMITED (FORMERLY EL TECH PRODUCTS AND TESTING) | FARIDABAD/INDIA | JAN 09, 2025 | JAN 08, 2027 |
| 89. | QAI/CIA/TL/2025/0097 | A L ENGINEERING SERVICES | GHAZIABAD/INDIA | JAN 16, 2025 | JAN 15, 2027 |
| 90. | QAI/CIA/TL/2025/0098 | MIGHTY TESTING PRIVATE LIMITED | GHAZIABAD/INDIA | JAN 23, 2025 | JAN 22, 2027 |
| 91. | QAI/CIA/TL/2025/0099 | ALPHA TESTING TECHNOLOGY PRIVATE LIMITED | GURUGRAM/INDIA | JAN 24, 2025 | JAN 23, 2027 |
| 92. | QAI/CIA/TL/2025/0100 | ALPHA TEST HOUSE PRIVATE LIMITED | BAHADURGARH/INDIA | JAN 30, 2025 | JAN 29, 2027 |
| 93. | QAI/CIA/TL/2025/0101 | MOLECULAR FORENSICS AND DNA TECHNOLOGIES (MFDT) | THIRUVANANTHAPURAM/INDIA | FEB 13, 2025 | FEB 12, 2027 |
| 94. | QAI/CIA/TL/2025/0102 | COTECNA INSPECTION INDIA PRIVATE LIMITED | INDORE/INDIA | FEB 13, 2025 | FEB 12, 2027 |
| 95. | QAI/CIA/TL/2025/0103 | EUROFINS BIOPHARMA PRODUCT TESTING INDIA PVT. LTD. | BENGALURU/INDIA | FEB 18, 2025 | FEB 17, 2027 |
| 96. | QAI/CIA/TL/2025/0104 | HINDUSTAN FORGING AND STEEL INDUSTRIES | THANE/ INDIA | FEB 25, 2025 | FEB 24, 2027 |
| 97. | QAI/CIA/TL/2025/0105 | STANDARD TESTING AND COMPLIANCE | FARIDABAD/INDIA | FEB 26, 2025 | FEB 25, 2027 |
| 98. | QAI/CIA/TL/2025/0106 | BRACHI TESTING SERVICES (BD) LTD. | GAZIPUR/BANGLADESH | MAR 03, 2025 | MAR 02, 2027 |
| 99. | QAI/CIA/TL/2025/0107 | METS LABORATORIES INDIA PRIVATE LIMITED | GURUGRAM/INDIA | MAR 07, 2025 | MAR 06, 2027 |
| 100. | QAI/CIA/TL/2025/0108 | PAAM TESTING LABS PRIVATE LIMITED | GREATER NOIDA/ INDIA | MAR 19, 2025 | MAR 18, 2027 |
| 101. | QAI/CIA/TL/2025/0109 | MITRA S.K. MOROCCO SARL | SARL, MOROCCO | MAR 29, 2025 | MAR 28 2027 |
| 102. | QAI/CIA/TL/2025/0110 | KSA LABS LLP | VADODARA/INDIA | Apr 07 ,2025 | Apr 06 , 2027 |
| 103. | QAI/CIA/TL/2025/0111 | VENUS TESTING & RESEARCH LABORATORY LLP (FORMERLY VENUS TESTING & RESEARCH LABORATORY) | RAIGARH/INDIA | APR 15, 2025 | APR 14, 2027 |
| 104. | QAI/CIA/TL/2025/0112 | EKO PRO ENGINEERS PRIVATE LIMITED | GHAZIABAD/INDIA | APR 22, 2025 | APR 21, 2027 |
| 105. | QAI/CIA/TL/2025/0113 | EKO PRO ENGINEERS PRIVATE LIMITED | GHAZIABAD/INDIA | APR 24,2025 | APR 23,2027 |
| 106. | QAI/CIA/TL/2025/0114 | BITUMEN TESTING LABORATORY | YANGON/ MYANMAR | MAY 03, 2025 | MAY 02, 2027 |
| 107. | QAI/CIA/TL/2025/0115 | PERFECT INTERNATIONAL TESTING LAB | GHAZIABAD/INDIA | MAY 06, 2025 | MAY 05, 2027 |
| 108. | QAI/CIA/TL/2025/0116 | ATCKON LABORATORIES LLP | BAHADURGARH,INDIA | MAY 06,2025 | MAY 05,2027 |
| 109. | QAI/CIA/TL/2025/0117 | GLOBAL SCIENTIC SOLUTION PRIVATE LIMITED | DELHI/INDIA | MAY 10, 2025 | MAY 09, 2027 |
| 110. | QAI/CIA/TL/2025/0118 | ESSQUARE GEO SERVICES LLP | VADODARA/INDIA | MAY 14, 2025 | MAY 13, 2027 |
| 111. | QAI/CIA/TL/2025/0119 | EUROFINS PRODUCT TESTING INDIA PRIVATE LIMITED | GURUGRAM/INDIA | MAY 14, 2025 | MAY 13 2027 |
| 112. | QAI/CIA/TL/2025/0120 | ATMY ANALYTICAL LABS PRIVATE LIMITED | FARIDABAD/INDIA | MAY 21, 2025 | MAY 20, 2027 |
| 113. | QAI/CIA/TL/2025/0121 | HOHENSTEIN INDIA PRIVATE LIMITED | GURUGRAM/INDIA | JUN 03, 2025 | JUN 02, 2027 |
| 114. | QAI/CIA/TL/2025/0122 | ASOPALAV MARITIME SERVICES PRIVATE LIMITED | KACHCHH/INDIA | JUN 10, 2025 | JUN 09 ,2027 |
| 115. | QAI/CIA/TL/2025/0123 | BUILDING RESEARCH LABORATORIES | YANGON/MYANMAR | JUN 18, 2025 | JUN 17, 2027 |
| 116. | QAI/CIA/TL/2025/0124 | KAILTECH TEST AND RESEARCH CENTRE PRIVATE LIMITED | INDORE/INDIA | JUN 25, 2025 | JUN 24, 2027 |
| 117. | QAI/CIA/TL/2025/0125 | AUN ANALYTICAL LABS PRIVATE LIMITED | NEW DELHI/INDIA | JUN 27, 2025 | JUN 26, 2027 |
| 118. | QAI/CIA/TL/2025/0126 | MARQUIS TECHNOLOGIES PRIVATE LIMITED | NOIDA/INDIA | JUN 27, 2025 | JUN 24, 2027 |
| 119. | QAI/CIA/TL/2025/0127 | INTERTEK INDIA PRIVATE LIMITED | TIRUPUR/INDIA | JUN 30, 2025 | JUN 29, 2027 |
| 120. | QAI/CIA/TL/2025/0128 | INTERTEK INDIA PRIVATE LIMITED | GURUGRAM/INDIA | JUL 02, 2025 | JUN 24, 2027 |
| 121. | QAI/CIA/TL/2025/0129 | RBD FIBERS LIMITED LABORATORY | NARAYANGANJ/BANGLADESH | JUL 12, 2025 | JUL 11, 2027 |
| 122. | QAI/CIA/TL/2025/0130 | PLANET ELECTRO LABS PRIVATE LIMITED | DELHI/INDIA | JUL 14 ,2025 | JUL 13, 2027 |
| 123. | QAI/CIA/TL/2025/0131 | CIMEC INFRA LABS PRIVATE LIMITED | GHAZIABAD/INDIA | JUL 21, 2025 | JUL 20, 2027 |
| 124. | QAI/CIA/TL/2025/0132 | BUREAU VERITAS CONSUMER PRODUCTS SERVICES (INDIA) PRIVATE LIMITED | TIRUPPUR/INDIA | JUL 29, 2025 | JUL 28, 2027 |
| 125. | QAI/CIA/TL/2025/0133 | IDMA LABORATORIES LIMITED | PANCHKULA/ INDIA | AUG 04, 2025 | AUG 03, 2027 |
| 126. | QAI/CIA/TL/2025/0134 | NCH SERVICES PRIVATE LIMITED | SURAT/INDIA | AUG 20, 2025 | AUG 19, 2027 |
| 127. | QAI/CIA/TL/2025/0135 | FIRE TEST & RESEARCH LABORATORY | SONIPAT, INDIA | Aug 20, 2025 | Aug 19, 2027 |
| 128. | QAI/CIA/TL/2025/0136 | ACCURATE TEST SOLUTIONS LLP | NOIDA/INDIA | Sep 05, 2025 | Sep 04, 2027 |
| 129. | QAI/CIA/TL/2025/0137 | KPBL SERVICES | GURUGRAM/INDIA | SEP 08, 2025 | SEP 07, 2027 |
| 130. | QAI/CIA/TL/2025/0138 | MEKANIKA TESTING LABS LLP | GHAZIABAD/INDIA | SEP 27, 2025 | SEP 26, 2027 |
| 131. | QAI/CIA/TL/2025/0139 | MANDALAY VETERINARY DIAGNOSTIC LABORATORY | MANDALAY/MYANMAR | SEP 29, 2025 | SEP 28, 2027 |
| 132. | QAI/CIA/TL/2025/0140 | SAMTEK TESTING LABORATORY LLP | LUCKNOW, INDIA | SEP 30, 2025 | SEP 29, 2027 |
| 133. | QAI/CIA/TL/2025/0141 | LABORATORIES FOR THE ENVIRONMENT, AGRICULTURE, AND FOOD (LEAF) | BEIRUT/ LEBANON | OCT 03, 2025 | OCT 02, 2028 |
| 134. | QAI/CIA/TL/2025/0142 | PLATFORM FOR RESEARCH AND ANALYSIS IN ENVIRONMENTAL SCIENCES (PRASE) | BEIRUT/ LEBANON | OCT 04, 2025 | OCT 03, 2028 |
| 135. | QAI/CIA/TL/2025/0143 | DMARA LABORATORIES - DMARA QUALITY SERVICES PRIVATE LIMITED | NAVI MUMBAI/INDIA | OCT 06, 2025 | OCT 05, 2027 |
| 136. | QAI/CIA/TL/2025/0144 | AADITECH TEST AND CALIBRATION LAB | DELHI/INDIA | OCT 10,2025 | OCT 09 ,2027 |
| 137. | QAI/CIA/TL/2025/0145 | TEKNOLAB QUALITY SERVICES PRIVATE LIMITED | FARIDABAD/ INDIA | OCT 11, 2025 | OCT 10, 2027 |
| 138. | QAI/CIA/TL/2025/0146 | BUREAU OF INDIAN STANDARDS, PATNA BRANCH LABORATORY | PATNA/ INDIA | OCT 14, 2025 | OCT 13, 2027 |
| 139. | QAI/CIA/TL/2025/0147 | HOHENSTEIN INDIA PRIVATE LIMITED | TIRUPPUR/INDIA | OCT 23, 2025 | OCT 22, 2027 |
| 140. | QAI/CIA/TL/2025/0148 | CHEMICAL LABORATORY | THIMPHU/BHUTAN | OCT 25, 2025 | OCT 24, 2027 |
| 141. | QAI/CIA/TL/2025/0149 | BAHRAIN FOOD AND WATER TESTING LABORATORY (UNIT OF BAHRAIN MEDICAL LAB) | KINGDOM OF BAHRAIN | NOV 12, 2025 | NOV 11, 2027 |
| 142. | QAI/CIA/TL/2025/0150 | SWOP ENGINEERING PRIVATE LIMITED | CHENNAI/INDIA | NOV 17, 2025 | NOV 16, 2027 |
| 143. | QAI/CIA/TL/2025/0151 | ALOK INDUSTRIES TESTING LABORATORY(WW) | VAPI/ INDIA | NOV 21, 2025 | NOV 20, 2027 |
| 144. | QAI/CIA/TL/2025/0152 | HETERO ANALYTICAL SOLUTION LLP | AHMEDABAD/INDIA | DEC 04, 2025 | DEC 03, 2027 |
| 145. | QAI/CIA/TL/2025/0153 | URS PRODUCTS AND TESTING PRIVATE LIMITED | NOIDA/ INDIA | DEC 10, 2025 | DEC 09, 2027 |
| 146. | QAI/CIA/TL/2025/0154 | JAWANDSONS TESTING LABORATORY | LUDHIANA/ INDIA | DEC 27, 2025 | DEC 26, 2027 |
| 147. | QAI/CIA/TL/2025/0155 | COTECNA INSPECTION INDIA PRIVATE LIMITED | BARBIL/ INDIA | DEC 28, 2025 | DEC 27, 2027 |
| 153. | QAI/CIA/TL/2026/0156 | SGS INDIA PRIVATE LIMITED | GURUGRAM/INDIA | JAN 07,2026 | JAN 06,2028 |
| 154. | QAI/CIA/TL/2026/0157 | GLOBAL PRODUCT STEWARDSHIP LABORATORY | MUMBAI/INDIA | JAN 20,2026 | JAN 19,2028 |
| 155. | QAI/CIA/TL/2026/0158 | GEO DYNAMIC ARABIA FOR SOIL INVESTIGATION AND MATERIAL TESTING | DUBA/SAUDIARABIA | JAN 23,2026 | JAN 22,2029 |
| 156. | QAI/CIA/TL/2026/0159 | COTECNA INSPECTION INDIA PRIVATE LIMITED | DHANBAD/INDIA | FEB 10,2026 | FEB 09,2028 |
| 157. | QAI/CIA/TL/2026/0160 | SHREE SHYAM JI HALLMARKING CENTRE (OPC) PRIVATE LIMITED | LUCKNOW/INDIA | FEB 16,2026 | FEB 15,2028 |
| 158. | QAI/CIA/TL/2026/0161 | ECO PARYAVRAN LABORATORIES AND CONSULTANTS PRIVATE LIMITED | MOHALI/INDIA | MAR 05, 2026 | MAR 04, 2028 |
| 159. | QAI/CIA/TL/2026/0162 | SMALL SCALE INDUSTRIES LABORATORY | YANGON/MYANMAR | MAR 19, 2026 | MAR 18, 2028 |
| 160. | QAI/CIA/TL/2026/0163 | REGIONAL FOOD RESEARCH AND ANALYSIS CENTRE | LUCKNOW/ INDIA | MAR 20, 2026 | MAR 19, 2028 |
| 161. | QAI/CIA/TL/2026/0164 | PRESTO LABORATORIES PRIVATE LIMITED | FARIDABAD/ INDIA | MAR 25, 2026 | MAR 24, 2028 |
| 162. | QAI/CIA/TL/2026/0165 | ELETTRA TECH LABS PRIVATE LIMITED | KANCHIPURAM/ INDIA | MAR 28, 2026 | MAR 27, 2028 |
| 163. | QAI/CIA/TL/2026/0166 | SGTL LABORATORY | NOIDA/ INDIA | APR 07, 2026 | APR 06, 2028 |
| 164. | QAI/CIA/TL/2026/0167 | MIDDLE EAST TESTING SERVICES BRANCH OF ABU DHABI 1 | ABU DHABI/ UNITED ARAB EMIRATES | APR 07, 2026 | APR 06, 2028 |
| S.N. | Accreditation Certificate No. | Name of | City/Country | Valid From | Valid until |
| 1. | QAI/CIA/TL/2023/0020 | SYSS LABS INDIA (FORMELY MDC TESTING & CERTIFICATIONS) | PANCHKULA/ INDIA | AUG 09, 2023 | AUG 08, 2025 |
| 2. | QAI/CLA/TL/2023/0016 | GENCHI GENBUTSU RESEARCH INSTITUTE (OPC) PVT. LTD. | AGRA/INDIA | JULY 21, 2023 | JULY 20, 2025 |
Calibration Laboratory Accreditation
| S.N. | Accreditation Certificate No. | Name of Accredited Laboratories | City/Country | Valid From | Valid until |
| 1. | QAI/CIA/CL/2023/0002 | AUTHENTIC INSTRUMENT & CALIBRATION SERVICES PVT. LTD | NOIDA/INDIA | MAR 29, 2025 | MAR 28, 2027 |
| 2. | QAI/CIA/CL/2024/0004 | MASTER LABS FOR TECHNICAL TESTING, INSPECTION AND CALIBRATION SERVICES | HIDD/ KINGDOM OF BAHRAIN | FEB 05, 2024 | FEB 04, 2026 |
| 3. | QAI/CIA/CL/2025/0005 | AADITECH TEST AND CALIBRATION LAB LLP | NEW DELHI/INDIA | OCT 06, 2025 | OCT 05, 2027 |
Biobanking Accreditation Programme
| S.N. | Accreditation Certificate No. | Name of Accredited Biobanks | City/Country | Valid From | Valid until |
| 1. | QAI/CIA/BB/2023/0002 | KARKINOS HEALTHCARE PRIVATE LIMITED BIOBANK | ERNAKULAM/INDIA | SEP 26, 2025 | SEP 25, 2027 |
| 2. | QAI/CIA/BB/2024/0004 | CBR BIOBANK, CENTRE FOR BRAIN RESEARCH, INDIAN INSTITUTE OF SCIENCE | BENGALURU/INDIA | AUG 08, 2024 | AUG 07, 2026 |
Inspection Bodies Accreditation
| S.N. | Accreditation Certificate No. | Name of Accredited Inspection Body | City/Country | Valid From | Valid until |
| 1. | QAI/CIA/IB/2025/0001 | Planet Electro Labs Private Limited- Inspection Division | NEW DELHI/INDIA | AUG 14, 2025 | AUG 13, 2027 |
Proficiency Testing Providers Accreditation
| S.No | Accreditation Certificate No. | Name of Accredited Proficiency Testing Providers | CITY/COUNTRY | VALID FROM | VALID UNTIL |
| 1 | QAI/CIA/PTP/2024/0001 | PT DIVISION - DMARA QUALITY SERVICES PRIVATE LIMITED | NAVI MUMBAI/INDIA | DEC 07, 2024 | DEC 06, 2026 |
| 2 | QAI/CIA/PTP/2025/0002 | NCH SERVICES PRIVATE LIMITED | SURAT/INDIA | NOV 18, 2025 | NOV 17, 2027 |
Reference Material Producers
| S.No | Accreditation Certificate No. | Name of Accredited Reference Material Producers | CITY/COUNTRY | VALID FROM | VALID UNTIL |
| 1 | QAI/CIA/RMP/2025/0001 | JALAN HALLMARKING CENTRE | DELHI/ INDIA | JUL 31, 2025 | JUL 30, 2027 |
| 2 | QAI/CIA/RMP/2025/0002 | NCH SERVICES PRIVATE LIMITED | SURAT/INDIA | AUG 26, 2025 | AUG 25, 2027 |
Medical Laboratory Recognition
List of Proficiency Testing Providers
Please click on the link given below to know the Proficiency Testing Providers
Recognition of QAI
National and International Regulators/Agencies Recognition/ Approval of QAI
1. Govt. Agencies/Regulators recognition/ approval for QAI Accreditation
Following but not limited to:
- Bureau of Indian Standards (BIS)
- Eastern Railway
- Central Public Works Department (CPWD)
- Power Finance Corporation (PFC)
- Legal Metrology, Department of Consumer Affairs
- Public Works Department, MP Government
- National Highway Authority of India (NHAI)
- Military Engineering Services (MES)
- Coal India Limited (CIL)
- Power Grid Corporation of India (PGCIL)
- Bureau of Energy Efficiency (BEE)
- BHEL Bhopal
- RITES Limited
- Directorate, Urban Administration and Development, MP, Bhopal
- PWD Bhopal
- Airport Authority of India
- Directorate General of Civil Aviation (DGCA)
- NBCC (India) Limited (A Government of India Enterprise)
- Atomic Energy Regulatory Board (AERB)
2. International Agencies/Regulators recognition/ approval for QAI Accreditation
- United States Consumer Products Safety Commission (US CPSC)
- United States Environmental Protection Agency (USEPA)-Formaldehyde Testing in wood/wood products
- World Organization of Animal Health (WOAH)
- Hongkong SAR for toy and consumer product testing as per Hongkong SAR SAR regulation
Adverse Decisions
| Sl.No. | Reference Number/ Certificate Number | Name of CAB, City/Country | Type of CAB | Reason for Adverse Decision | Decision & Date |
| 1 | QAI/CIA/CL/2023/0003 | NEUTECH AUTOMATION | CALIBRATION | Clause number 11.c of QAI CIA 022 (Non-payment of accreditation expenses like assessment or re-assessment charges and annual accreditation fees) | SUSPENDED W.E.F. 26 MARCH 2025 |
| 2 | QAI/CIA/TL/2024/0075 | SHRI BAJRANG TESTING LABS, GREATER NOIDA/INDIA | TESTING | Clause number 4.2 d of QAI CIA 022 (Submitting a fake QAI accreditation certificate to BIS) | Closed and put under debarred category w.e.f. 16 May 2024 |
| 3 | QAI/CLA/ML/2023/0016 | SYNERGY CARE CLINIC , MALDIVES | MEDICAL TESTING | Clause number 5.4 of QAI CIA 022 (CAB has not submitted the documents for the ongoing monitoring & has not paid the Accreditation fees, beyond three months of the due date) | SUSPENDED W.E.F 05 JULY 2025 |
| 4 | QAI/CLA/TL/2023/0019 | LEGION LABORATORIES PVT. LTD., GREATER NOIDA/INDIA | TESTING | Clause number 5.4 of QAI CIA 022 (CAB has not submitted the documents for the ongoing monitoring & has not paid the Accreditation fees, beyond three months of the due date) | SUSPENDED W.E.F. 26 MARCH 2025 |
| 5 | QAI/CLA/TL/2023/0040 | BIOMEDICA LABORATORIES, INDORE/INDIA | TESTING | Clause number 4.1 d of QAI CIA 022 (CAB is found with fraudulent behaviour, intentionally provide false information, conceals information that is required as per the process) | Closed w.e.f. 9 January 2024 |
| 6 | QAI/CLA/ML/2019/0007 | DEPARTMENT OF CLINICAL BIOCHEMISTRY, SHER-I-KASHMIR INSTITUTE OF MEDICAL SCIENCES | MEDICAL | Clause number 5.4 of QAI CIA 022 (CAB has not submitted the documents for the ongoing monitoring & has not paid the Accreditation fees, beyond three months of the due date) | SUSPENDED W.E.F. 13 NOVEMBER 2025 |
Other Decisions
| SL.NO. | REFERENCE NUMBER | NAME OF THE CAB, CITY/COUNTRY | TYPE OF CAB | VALIDITY OF ACCREDITATION | DECISION & DATE |
| 1 | QAI/CIA/BB/2023/0003 | BIOREPOSITORY FACILITY, TRANSLATIONAL HEALTH SCIENCE AND TECHNOLOGY INSTITUTE,FARIDABAD/ INDIA | BIOBANK | FROM DEC 09, 2023 UNTIL DEC 08, 2025 | VOLUNTARY WITHDRAWAL W.E.F NOV 26, 2025 |
| 2 | QAI/CIA/CL/2022/0001 | S.V. INDIA CALIBRATION SERVICES, RUDRAPUR/INDIA | CALIBRATION | FROM JAN 04, 2023 UNTIL 03 JAN, 2025 | VOLUNTARY WITHDRAWAL W.E.F FEB 23, 2024 |
| 3 | QAI/CIA/ML/2023/0019 | SALVUS BIORESEARCH SOLUTIONS,AHMEDABAD | MEDICAL | FROM DEC 23, 2023- UNTIL Dec 22,2025 | VOLUNTARY WITHDRAWAL W.E.F JAN 10,2025 |
| 4 | QAI/CIA/TL/2023/0035 | BIOZED ENGINEERING PRIVATE LIMITED, THANE/INDIA | TESTING | FROM DEC 22, 2023 UNTIL DEC 21, 2025 | VOLUNTARY WITHDRAWAL W.E.F. 01 NOVEMBER 2025 |
| 5 | QAI/CIA/TL/2024/0044 | RF TR LAB-TARANG LABS, WIPRO LIMITED, | BENGALURU/INDIA | FROM FEB 19, 2024 UNTIL FEB 18, 2026 | VOLUNTARY WITHDRAWAL W.E.F. 03 MARCH 2025 |
| 6 | QAI/CLA/BB/2021/0001 | NATIONAL LIVER DISEASE BIOBANK, INSTITUTE OF LIVER & BILIARY SCIENCES, NEW DELHI/ INDIA | BIOBANK | FROM SEP 28, 2021 UNTIL SEP 27, 2023 | DEC 27, 2023 |
| 7 | QAI/CLA/ML/2019/0006 | CHEMBAKASSERY COMPUTERISED LAB, PUDUKAD/KERALA | MEDICAL | FROM AUG 04, 2019 UNTIL AUG 03, 2021 | VOLUNTARY WITHDRAWAL W.E.F MAR 17, 2021 |
| 8 | QAI/CLA/ML/2019/0008 | MEDILAB SPECIALITY LABORATORIES PVT. LTD., KOCHI/INDIA | MEDICAL | FROM AUG 15, 2019 UNTIL AUG 14, 2021 | VOLUNTARY WITHDRAWAL W.E.F MAR 17, 2021 |
| 9 | QAI/CLA/TL/2018/0001 | DATT MEDIPRODUCT PVT. LTD., NUH/INDIA | TESTING | FROM NOV 26, 2022 UNTIL NOV 25, 2024 | VOLUNTARY WITHDRAWAL W.E.F 17 FEBRUARY 2024 |
| 10 | QAI/CLA/TL/2018/0001 | DEPARTMENT OF LIFE SCIENCES (R&D), DATT MEDIPRODUCTS PVT. LTD., NUH/INDIA | TESTING | FROM NOV 26, 2022 UNTIL NOV 25, 2024 | VOLUNTARY WITHDRAWAL W.E.F.17 FEBRUARY 2024 |
| 11 | QAI/CLA/TL/2022/0004 | MDC TESTING AND CERTIFICATIONS, MOHALI/INDIA | TESTING | FROM FEB 22, 2022 UNTIL MAY 01, 2023 | ADDRESS CHANGE, NEW CERTIFICATE ISSUED- QAI/CLA/TL/2023/0020 |
| 12 | QAI/CLA/TL/2023/0007 | AGSS ANALYTICAL AND RESEARCH LAB PRIVATE LIMITED | TESTING | FROM MAR 25, 2023 UNTIL MAR 24, 2025 | VOLUNTARY WITHDRAWAL W.E.F DEC 28, 2024 |
| 13 | QAI/CLA/TL/2023/0007 | AGSS ANALYTICAL AND RESEARCH LAB PVT. LTD., NEW DELHI/INDIA | TESTING | FROM MAR 25, 2023 UNTIL MAR 24, 2025 | VOLUNTARY WITHDRAWAL W.E.F. 23 DECEMBER 2024 |
FAQ’s About The Service
There are many questions about the service, we have selected frequently asked questions about this service. If you do not see your answer, please contact us.
1. What is the primary objective of the Centre for International Accreditation (CIA)?
2. Which types of CABs (Conformity Assessment Bodies) can apply for CIA accreditation?
3. Is CIA accreditation available only to CABs in India?
4. How is the CIA accreditation process governed and implemented?
Services
- About CIA
- Board
- Organisation Structure
- General Documents
- Medical Laboratory Accreditation
- Testing Laboratory Accreditation
- Calibration Laboratory Accreditation
- Biobanking Accreditation Programme
- Inspection Bodies Accreditation
- PT Provider Accreditation
- Reference Material Producers Accreditation
- Medical Laboratory Recognition
- List of Proficiency Testing Providers
- Recognition of QAI
- Adverse Decisions
- Other Decisions










